NIH style 4 cohort study with 400 patients total sample size
CLINICAL TRIAL PROTOCOL (NIH-STYLE SUBMISSION)
Project Title
NAD+ Augmentation to Improve Metabolic, Cardiac, and Neurocognitive Function: A Randomized Controlled Trial Across High-Risk Adult Cohorts
Principal Investigators
Tontania English RN – registered nurse
Kalki Bommaraju MD MPH – internal medicine doctor
Philip F Binkley MD MPH – cardiologist



Experience
Clinical management of patients with metabolic disease, heart failure, and complex chronic illness
Experience in inpatient and outpatient medicine
Interest in translational physiology and systems-level disease mechanisms
Personal Statement
Our clinical experience has emphasized the limitations of disease-specific treatment models and the need for therapies targeting shared underlying biologic mechanisms. This proposal represents a translational approach bridging NAD+ biology with clinical outcomes.
Project Summary / Abstract
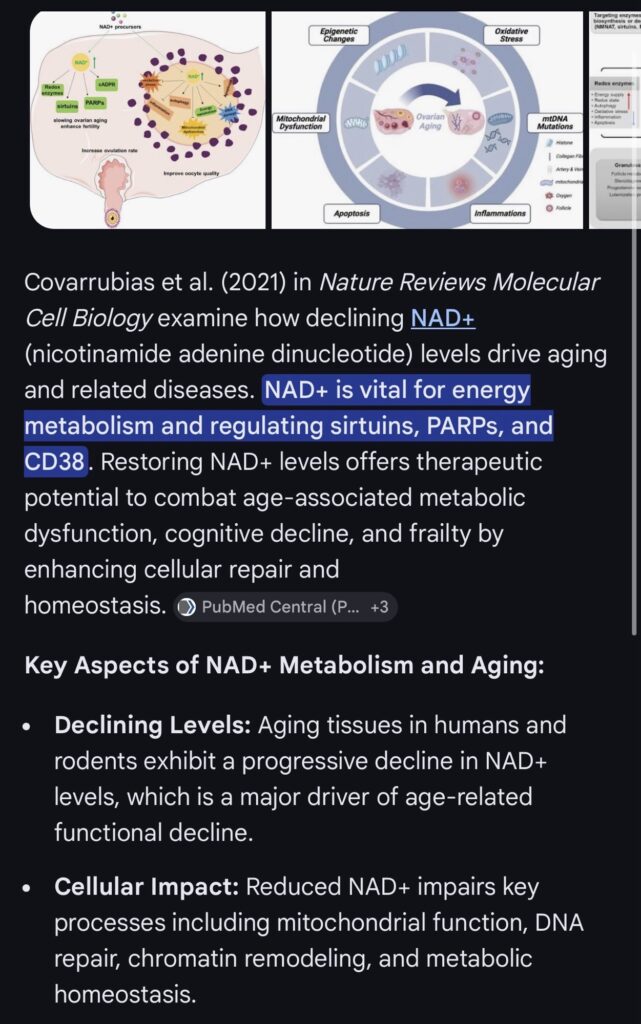
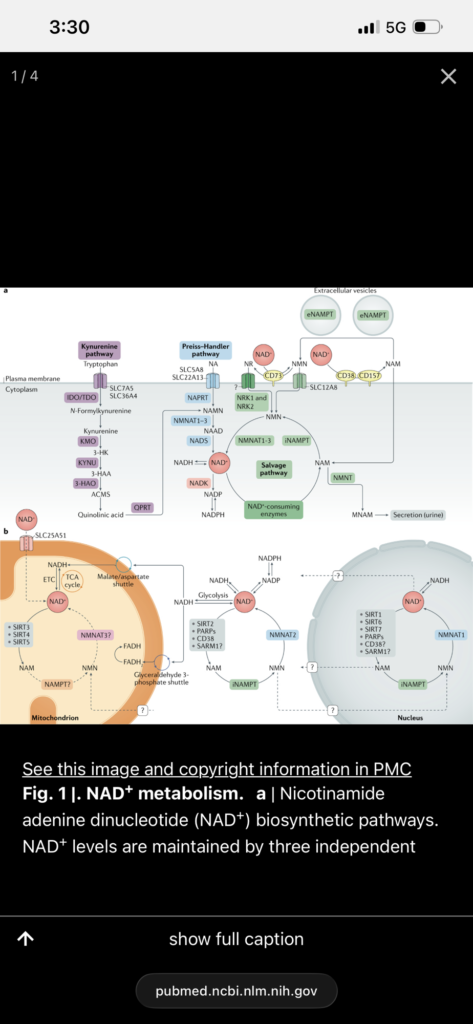
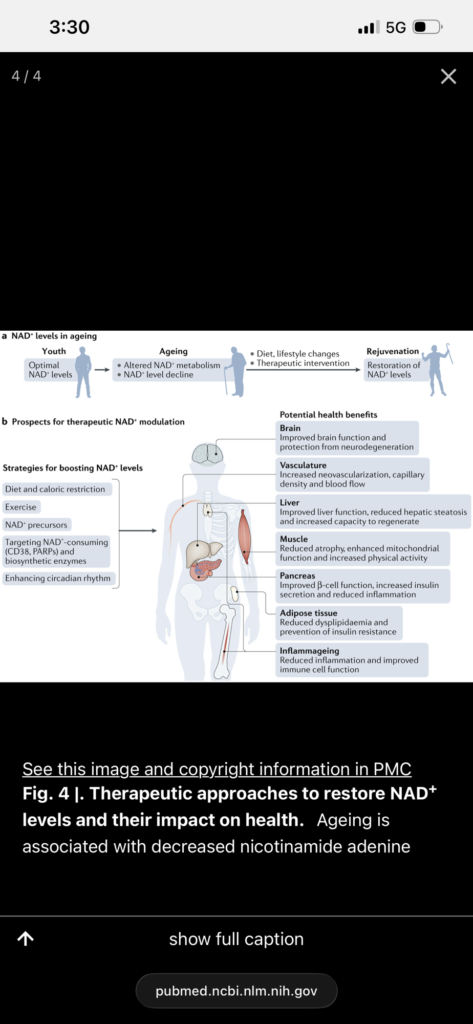
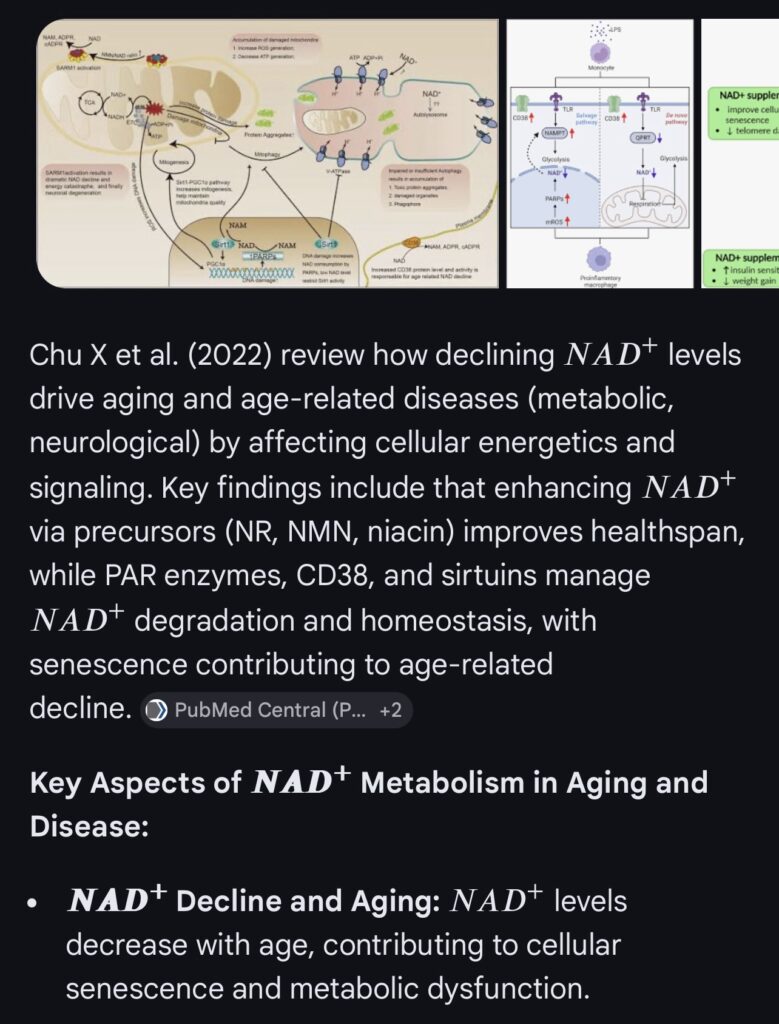
Age-related decline in NAD+ is associated with mitochondrial dysfunction, impaired metabolic regulation, and reduced cellular resilience. Preclinical studies demonstrate that restoration of NAD+ improves metabolic, cardiac, and neurocognitive function. However, rigorous randomized controlled trials in humans remain limited.
This study proposes a multi-cohort, randomized, double-blind, placebo-controlled trial evaluating NAD+ augmentation in four high-risk populations: prediabetes, early dementia, heart failure, and obesity. Each cohort targets a clinically meaningful condition with validated endpoints while testing a shared mechanistic hypothesis that restoration of NAD+ improves mitochondrial and metabolic function across organ systems.
A total of 400 participants will be enrolled, with 100 participants in each cohort. Within each cohort, participants will be randomized 1:1 to receive NAD+ infusion or placebo for 6 months, with follow-up through 12 months. Primary outcomes are cohort-specific and include insulin sensitivity, cognitive performance, functional capacity, and body composition. Secondary outcomes include mitochondrial biomarkers, physiologic monitoring, and NAD+ levels.
This study will provide the first rigorous evaluation of NAD+ augmentation across multiple disease domains and determine whether targeting a core metabolic pathway yields clinically meaningful benefit.
Specific Aims
Overall Hypothesis
Restoration of NAD+ improves mitochondrial and metabolic function, leading to measurable improvements in disease-specific outcomes across multiple high-risk populations.
Aim 1
Determine whether NAD+ augmentation improves insulin sensitivity in adults with prediabetes.
Primary Outcome: Change in HOMA-IR and glucose tolerance.
HOMA-IR stands for Homeostatic Model Assessment of Insulin Resistance.
It is a method used to estimate how resistant a person is to insulin using fasting laboratory values.
What it measures
It estimates how much insulin the body needs to maintain normal blood glucose levels.
Higher insulin levels relative to glucose suggest insulin resistance.
Lower insulin levels with normal glucose suggest good insulin sensitivity.
Formula
HOMA-IR = (Fasting Insulin × Fasting Glucose) / 405
Fasting insulin is measured in micro units per milliliter (µU/mL).
Fasting glucose is measured in milligrams per deciliter (mg/dL).
Example
If fasting glucose is 100 and fasting insulin is 10:
HOMA-IR = (10 × 100) / 405 = approximately 2.47
Interpretation (approximate ranges)
Around 1.0 indicates normal insulin sensitivity
1.5 to 2.0 suggests early insulin resistance
Greater than 2.5 suggests insulin resistance
Greater than 3.0 suggests significant insulin resistance
Ranges can vary depending on the population and laboratory standards.
Why it is used
HOMA-IR is commonly used in clinical studies because it is simple, inexpensive, and can be repeated over time to track changes in metabolic function.
Limitation
It is an estimate and not a direct measurement.
The gold standard for measuring insulin sensitivity is the hyperinsulinemic-euglycemic clamp, which is more complex and resource-intensive.
Aim 2
Determine whether NAD+ augmentation improves cognitive function in adults with early dementia.
Primary Outcome: Change in standardized cognitive scores (MoCA, ADAS-Cog).
MoCA stands for Montreal Cognitive Assessment.
It is a screening test used to detect mild cognitive impairment and early dementia.
What it measures
It evaluates multiple areas of cognition, including attention, memory, language, executive function, visuospatial ability, and orientation.
Structure
The test is scored out of 30 points and takes about 10 minutes to complete.
Interpretation
A score of 26 to 30 is considered normal
A score below 26 suggests cognitive impairment
Lower scores indicate greater impairment
Why it is used
MoCA is sensitive for detecting early cognitive decline and is commonly used in clinical studies because it can detect subtle changes over time.
ADAS-Cog stands for Alzheimer’s Disease Assessment Scale – Cognitive Subscale.
It is a more detailed cognitive test used primarily in Alzheimer’s disease research and clinical trials.
What it measures
It evaluates memory, language, attention, and other cognitive functions in more detail than screening tests.
Structure
The score typically ranges from 0 to 70 depending on the version used.
Interpretation
Lower scores indicate better cognitive function
Higher scores indicate worse cognitive function
An increase in score over time suggests worsening cognition
A decrease in score suggests improvement
Why it is used
ADAS-Cog is widely used in clinical trials because it is sensitive to changes in cognitive performance over time, especially in patients with established dementia.
Key difference
MoCA is a short screening tool used to detect early cognitive changes, and higher scores are better.
ADAS-Cog is a more detailed assessment used in research settings, and lower scores are better.
Why both are used in studies
Using both tests provides a more complete evaluation of cognition. MoCA helps detect early or subtle changes, while ADAS-Cog measures more detailed changes over time.
Aim 3
Determine whether NAD+ augmentation improves functional capacity in patients with heart failure.
Primary Outcome: Change in 6-minute walk distance and NT-proBNP.
Aim 4
Determine whether NAD+ augmentation improves metabolic health in adults with obesity.
Primary Outcome: Change in body composition (DEXA) and insulin sensitivity.
Secondary Aim
Evaluate changes in mitochondrial function, NAD+ levels, inflammatory markers, and physiologic stability across all cohorts.
Research Strategy
Significance
Chronic diseases including diabetes, dementia, heart failure, and obesity share upstream mechanisms involving mitochondrial dysfunction and impaired cellular energetics. Current therapies primarily target disease-specific pathways rather than underlying metabolic dysfunction.
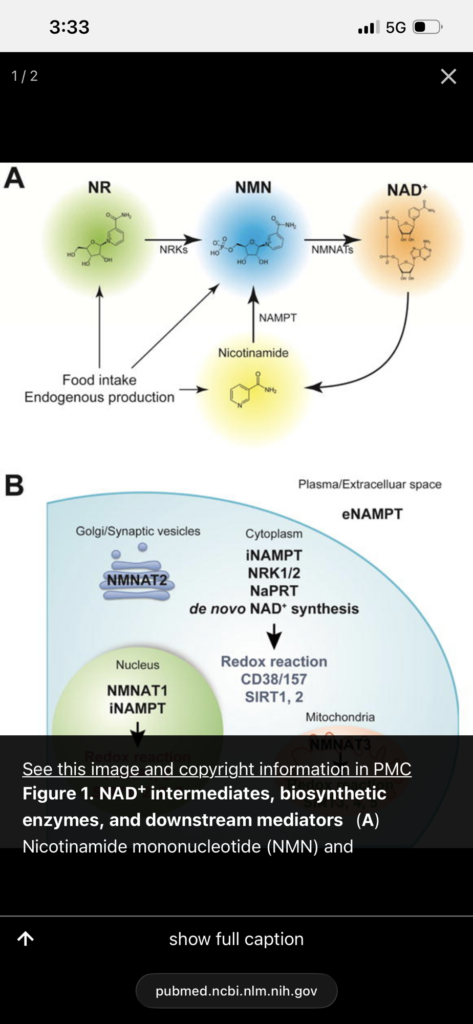
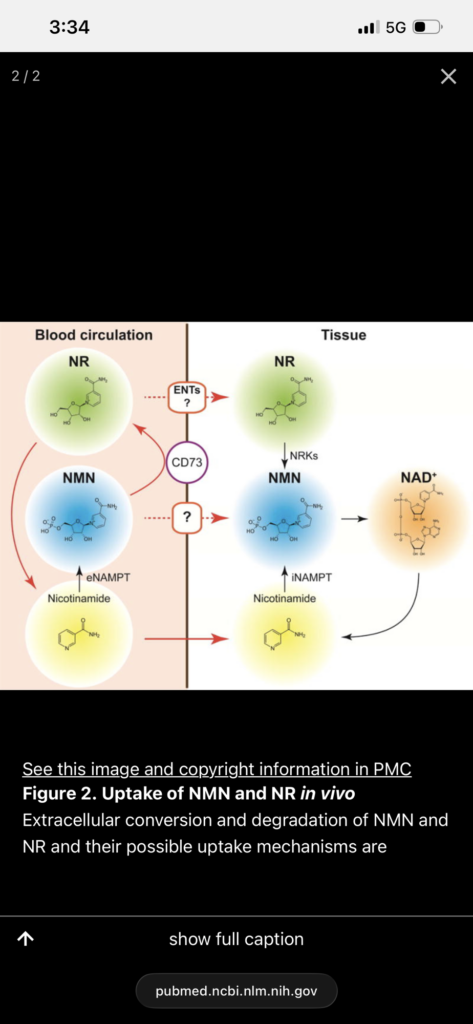
NAD+ is a central regulator of mitochondrial function, DNA repair, and metabolic signaling. Declining NAD+ levels are associated with aging and multiple disease states. Restoration of NAD+ improves metabolic and functional outcomes in preclinical models, but clinical evidence in humans is limited.
This study addresses a critical gap by testing whether NAD+ augmentation produces clinically meaningful improvements across multiple disease domains using a rigorous randomized controlled design.
Innovation
This study is innovative because it targets a shared upstream metabolic pathway across multiple diseases rather than isolated downstream processes. It represents the first randomized controlled evaluation of intravenous NAD+ across multiple clinical populations. It integrates metabolic, cognitive, and functional endpoints into a unified design and incorporates continuous physiologic monitoring for exploratory biomarker discovery.
Approach
Study Design
Multi-cohort, randomized, double-blind, placebo-controlled trial with four parallel cohorts: prediabetes, early dementia, heart failure (NYHA class II–III), and obesity. Participants are randomized 1:1 within each cohort.
Sample Size
Total n = 400 participants
100 participants per cohort
50 NAD+ and 50 placebo per cohort
Sample size is powered to detect clinically meaningful differences in primary outcomes within each cohort.
Population
Inclusion Criteria
Age 40–70
Diagnosis specific to cohort (prediabetes, early dementia, heart failure, or obesity)
Ability to provide informed consent
Exclusion Criteria
Contraindications to infusion therapy
Intervention
Experimental Group
Intravenous NAD+ infusion administered weekly for 12 weeks followed by maintenance dosing per protocol
Control Group
Placebo infusion (normal saline) with identical administration schedule
Primary Outcomes
Prediabetes
Insulin sensitivity (HOMA-IR or hyperinsulinemic-euglycemic clamp)
Dementia
Cognitive performance (MoCA, ADAS-Cog)
Heart Failure
6-minute walk distance
NT-proBNP levels
Obesity
Body composition (DEXA)
Insulin sensitivity
Secondary Outcomes (All Cohorts)
NAD+ levels
Inflammatory markers
VO2 max
Resting energy expenditure
Quality of life measures
Exploratory Outcomes
Continuous glucose monitoring
Heart rate variability
Wearable device data
AI-based physiologic pattern analysis
Data Collection
Baseline
Metabolic laboratory testing
Functional testing
Body composition analysis
NAD+ level measurement
Follow-Up
Monthly laboratory monitoring
Functional testing at 3 and 6 months
Continuous wearable monitoring
Final assessment at 12 months
Statistical Analysis
Intention-to-treat analysis
Mixed-effects models for repeated measures
Between-group comparisons within each cohort
Secondary pooled analyses across cohorts
Adjustment for baseline covariates
Potential Problems and Alternative Strategies
If effect sizes are smaller than expected, sensitive endpoints and subgroup analyses will be used. If heterogeneity is observed across cohorts, analyses will remain cohort-specific with secondary pooled exploratory analysis.
Timeline
Year 1: Study setup and enrollment
Year 2: Intervention and data collection
Year 3: Data analysis and publication
Investigator Biosketch (Abbreviated)
Name: Tontania English, RN
Kalki Bommaraju MD MPH
Philip F Binkley MD MPH
Role: Principal Investigators
Training
Registered Nurse
Doctor of Medicine, Internal Medicine
Cardiologist
Experience
Clinical management of patients with metabolic disease, heart failure, and complex chronic illness
Experience in inpatient and outpatient medicine
Interest in translational physiology and systems-level disease mechanisms
Personal Statement
Our clinical experience has emphasized the limitations of disease-specific treatment models and the need for therapies targeting shared underlying biologic mechanisms. This proposal represents a translational approach bridging NAD+ biology with clinical outcomes.
Budget (Estimated, 3-Year Project)
Total estimated budget: 3.0 to 3.8 million USD
Major cost categories include personnel, infusion services, laboratory assays, imaging (DEXA), cognitive testing, wearable monitoring devices, and data management systems.
Overall Impact
This study directly tests whether restoring NAD+ levels can modify fundamental metabolic and physiologic processes across multiple disease states. Positive findings would support a new therapeutic framework targeting shared mechanisms of aging and chronic disease and justify larger multicenter trials.
Significance (Additional Rationale: Real-World Use and Evidence Gap)
NAD+ intravenous infusion therapy is increasingly being offered through a rapidly expanding number of outpatient infusion centers across the United States. These services are being marketed directly to consumers with claims related to improved energy, cognitive function, metabolic health, and aging-related outcomes. Despite widespread adoption, there is a lack of rigorous clinical trial data evaluating the safety, efficacy, optimal dosing, and appropriate patient selection for this intervention.
As a result, a growing number of patients are receiving NAD+ infusions in real-world settings without an established evidence base to guide clinical decision-making. This creates a critical gap between clinical practice and scientific validation. In the absence of controlled studies, clinicians, patients, and regulatory bodies lack reliable data regarding therapeutic benefit, potential risks, and long-term outcomes.
The widespread, unsupervised use of NAD+ infusion therapy underscores the urgency of conducting a well-designed randomized controlled trial. Such a study is necessary not only to evaluate efficacy, but also to define safety profiles, identify populations most likely to benefit, and establish standardized protocols for administration and monitoring.
This proposal directly addresses this gap by providing a rigorous, controlled evaluation of NAD+ augmentation across multiple clinically relevant populations. Given the current scale of real-world use, generating high-quality evidence is essential to inform clinical practice, guide patient care, and support evidence-based policy decisions.